Our Approach
Miramax Clinical Research, is a multi-specialty clinical research center who conducts clinical trials from Phase I to Phase IV. These studies involve a variety of areas but not limited to:
- Cardiology
- Neurology
- Psychiatry
- Internal Medicine
- Dermatology
- Endocrinology
- Infectious Disease
- Rheumatology
- Gynecology
- Pulmonology
- Podiatry
- Infectious Disease
- COVID-19
Our Story
Is a privately owned clinical research site located directly in front of Jackson North Medical Center/Hospital & Emergency Department.
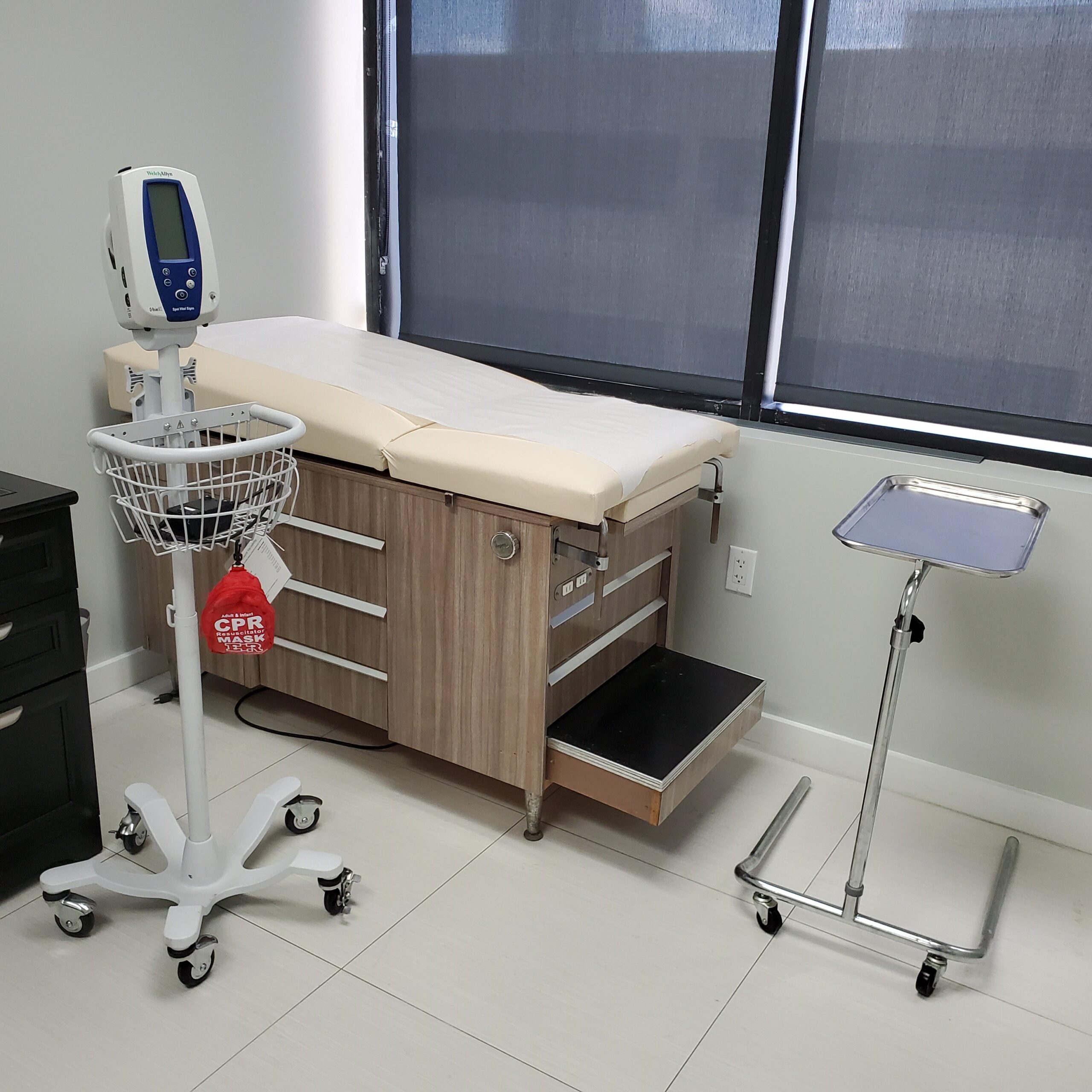
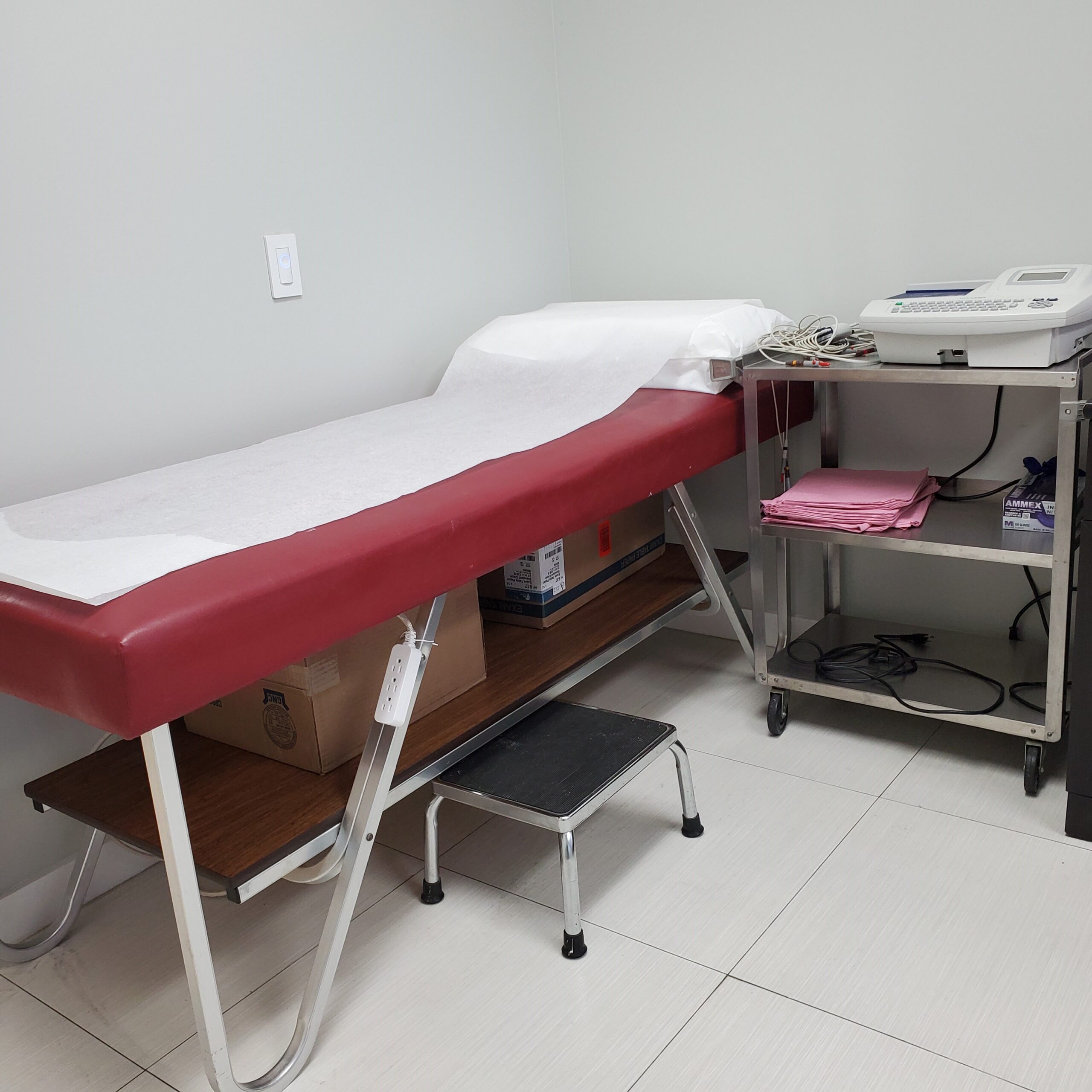
Our Facility
- Emergency Crash Cart (Revised December 2022)
- Cardiac Defibrillator
- We use cutting-edge eSource & CTMS technology software for the outmost efficiency in recording data from our clinical research Trials.
- Our facility is climate-controlled and fully equipped with a state of the art conference room, complete with wireless internet access, VoIP, HD Teleconference, printer, copier and fax capabilities.
- Dedicated area for storage of study binders with locked and limited access.
- Locked and limited access drug and supply storage facility.
- Locked -20º temperature monitored freezer.
- Locked -80º temperature monitored freezer.
- Locked medical grade temperature monitored refrigerator.
- Local laboratory, centrifuges, ECG Machine, CPAP, Portable Oxygen, Nebulizers, Spirometry with DLCO
- In house High-End Diagnostic Ultrasound for: Echocardiography (Tissue Doppler, TAPSE), Transvaginal Sonography, Etc.
- Access to Dry Ice.
- Patient rest area.
- Easy access to multiple hotels less than one mile.
- Ample parking space and a comfortable waiting area.
- We are CLIA certified.
- OSHA Compliant
- HIPAA trained staff in research guidelines.
- IATA trained.
- GDUFA self-identified.
- Point of Care Infusion capabilities via pump, syringe and PCA for the delivery of fluids, medications, blood, and blood products using continuous or intermittent delivery through clinically acceptable routes of administration such as:
- Intravenous (IV)
- Intra-arterial (IA)
- Subcutaneous
- Epidural
- Enteral
Meet the Team
We have a very special and dedicated group and our philosophy is to process each and every study with detailed attention and responsibility.
Why? Because people want better health! It's a human thing.
Winston Manga
Site Director / Regulatory Officer
Guadalupe Rengifo
Business Development / Project Manager
Bibiana Borrero
Clinical Research Coordinator
Nelmis Diaz
Clinical Research Coordinator
Lazaro Diaz
Clinical Research Coordinator
Jared M. Lechtenstein, D.O.
Principal Investigator
Armando Curra, M.D.
Principal Investigator
Elias Mualin, M.D.
Principal Investigator
Tessey Jose, M.D.
Principal Investigator
David Jassir, M.D.
Principal Investigator
Lucky Flores, M.D.
Principal Investigator
Francisco Bautista-Mena, M.D.
Principal Investigator
George Rovito, M.D.
Principal Investigator
Christine Jassir, ARNP
Sub Investigator
Helen Nau, ARNP
Sub Investigator
Next Steps...
To participate in a clinical trial please call us at (954) 363-1163